This article was originally published on Nature’s Rainbow.
Seems to go without saying for most experienced fluorescent mineral collectors; rocks that sit exposed to the elements for millennia (give or take a few million years) develop a “rind” on the outside, oxidize, and weather. Often there is no suggestion of fluorescence inside, or maybe just a little hint. Exposure to the elements will, in many cases, kill fluorescence.
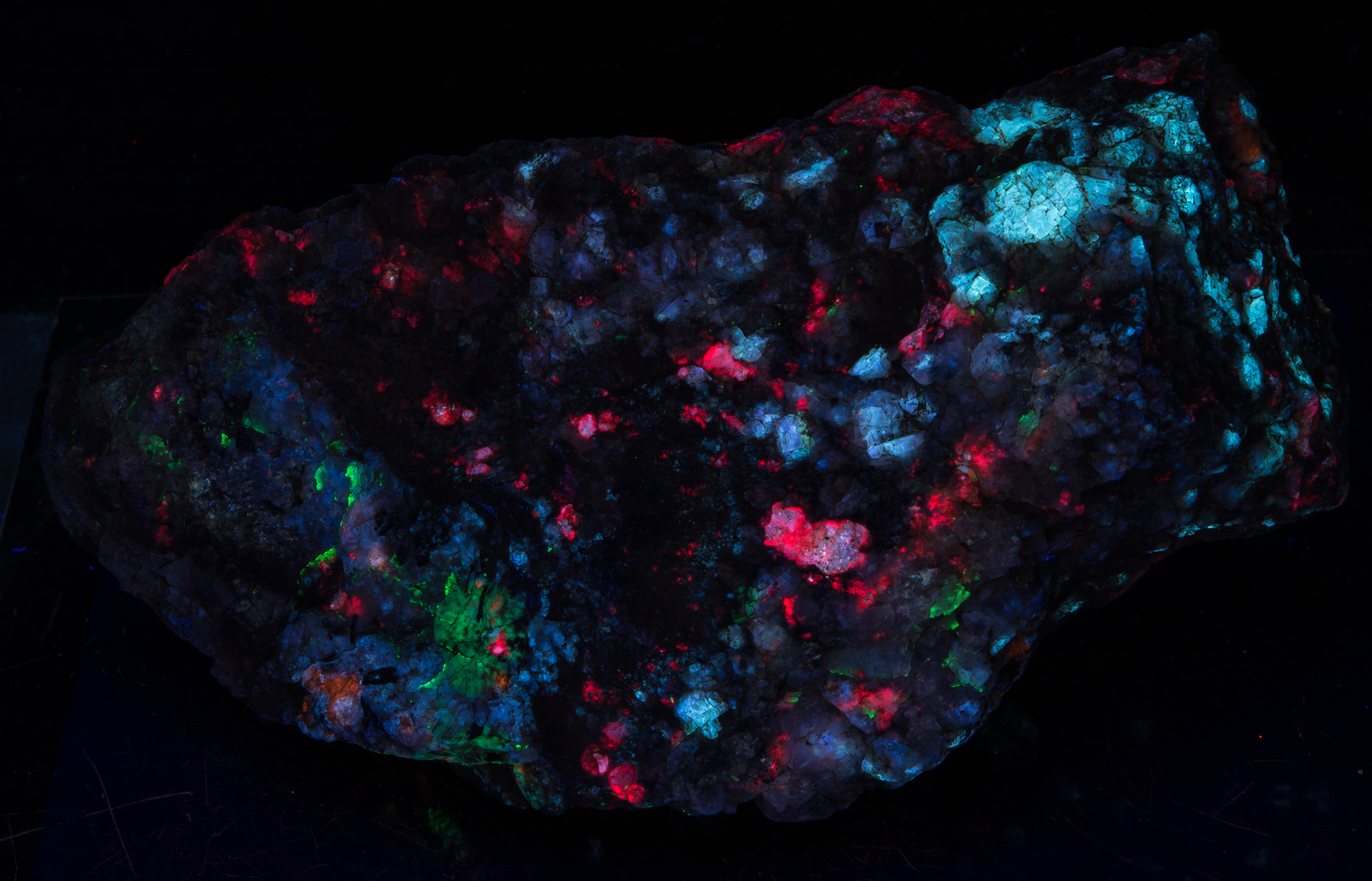
Crack it! This has been my mantra for years, told to new folks I’ve guided into the mountains of Greenland. For ten years I guided groups of 5 to 7 people into the mountains and put them on the best collecting spots in the Ilimaussaq Complex. They would start collecting rocks off the ground, throwing them in their backpacks if they even showed a hint of fluorescence (the typical new visitor wanted to be sure they weren’t skunked, and had no idea of the wonders they would find once they gained a little experience). Often I found they were not even cracking rock and were leaving pieces that had a lot of potential just laying there. At the end of the day they carried their packs off the mountain, back to the hostel, and unloaded. Invariably the rocks from these first few days were culled out at the end of the tour as junk. And even though they busted their butts carrying them off the mountain, they didn’t bother splitting them to see what was inside. They left ’em behind in Greenland – what a waste of blood, sweat, and even some tears. (I, on the other hand, figured if they thought that rock was good enough to carry off the mountain I might as well throw it in our shipping container and send it back to TN.)
This is one such rock. A few days ago I was down at the barn splitting rock with a friend. We came across this piece (half buried in the dirt) that looked interesting from the outside but clearly was no winner (first pic). I put it in my rock splitter and it graciously split right down the middle, yielding two nice halves with a third respectable piece.