Originally posted on Nature’s Rainbows in 2018 by Chris Clemens.
…at least not in terms of fluorescence. As is the case with many fluorescent minerals, different specimens of the same mineral species can show tremendous variability in fluorescent response, even among those specimens collected from the same location. This is especially the case for those minerals that are dependent on the inclusion of extrinsic activators as part of their mechanism of fluorescence. The mineral adamite provides an excellent example of this phenomenon.
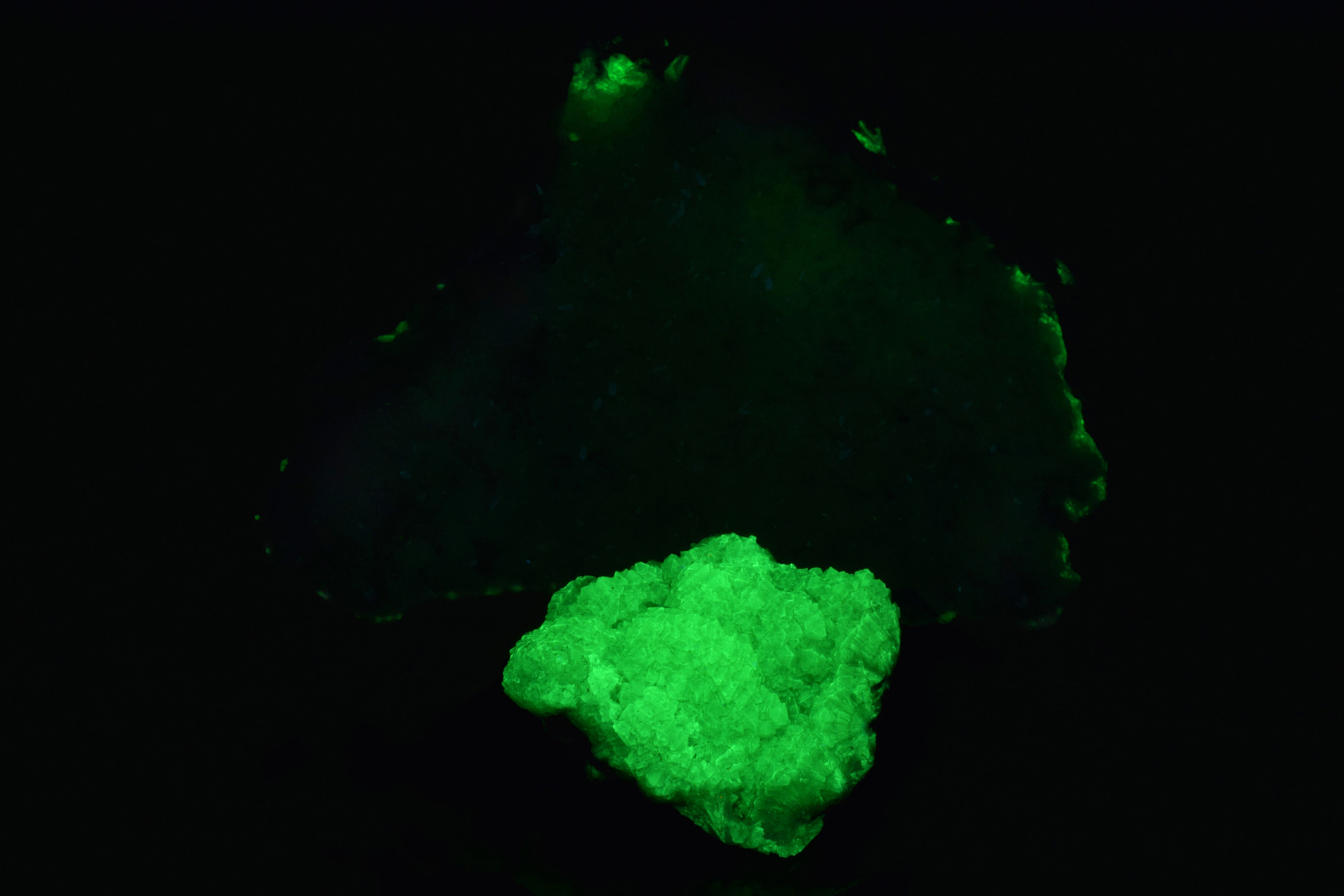
Adamite is a secondary zinc arsenate hydroxide found in zinc ore deposits containing arsenic-bearing minerals. One of the premier adamite locations is the Ojuela Mine, located in Mapimi, Durango, Mexico. Adamite from Mapimi often forms as attractive lime green crystals on a contrasting matrix of reddish/brown limonite, and is a highly desirable collector species due to its beautiful aesthetics. In addition to providing beautiful visible light display specimens, the adamite from Mapimi is also of interest to the collector of fluorescent minerals due to the brilliance of its green fluorescence under short wave UV. The fluorescent brightness of some Mapimi adamites rivals that of Franklin willemite. The green fluorescence in adamite is caused by the incorporation of minor amounts of an extrinsic activator, the uranyl ion [(UO2)2+], into the crystal structure of the adamite, causing classic “uranyl-activated” fluorescence under UV. However, as shown in the following example, the fluorescent response of Mapimi adamite is highly variable from specimen to specimen.
The following photograph shows two specimens of adamite from Mapimi, Mexico, as seen under visible light. One smaller specimen in the foreground, and a larger specimen in the background:
I thought it would be interesting to conduct a quick experiment to determine a potential explanation for the difference in fluorescence between these two specimens. Using a Geiger counter (Ludlum Model 3 Survey Meter) I measured the radioactivity emitted by each of these two specimens to obtain an estimate of the relative uranium content of each.
Other factors may also influence the fluorescence of adamite. Iron is a known quencher of fluorescence, and is certainly present in the red/brown limonitic gossan matrix of these Mapimi adamites. Perhaps trace amounts of iron present in the adamite mineral structure also affects fluorescence.
These results are not definitive due to the very small sampling size; however, they do suggest that the uranyl content of Mapimi adamites is variable, and that higher uranyl percentages correlate with a brighter fluorescent response under UV. In the end, all adamites are not created equal. At least not in terms of fluorescent response. The purchase of a choice adamite specimen from the classic Mapimi, Mexico, location is no guarantee of a killer fluorescent specimen.