Aragonite – Sicily, Italy
Contributed by: Michael Crawford
Date: Apr 17th, 2026
Locality: Giumentaro Mine, Capodarso, Enna, Enna Province, Sicily, Italy (See on Mindat)
Size: 7 x 12 cm
Description:
A cluster of hexagonal pale green aragonite crystals from Giumentaro Mine, Enna Province, Sicily, Italy.
The fluorescent color changes with the wavelength of UV illumination. It fluoresces pinkish red under longwave light, pink under midwave light, and bluish white under SW light. The specimen also has long-lasting phosphorescence/afterglow from exposure to all wavelengths of UV light. The color of the afterglow is green from longwave UV exposure and blue from midwave and shortwave UV exposure.
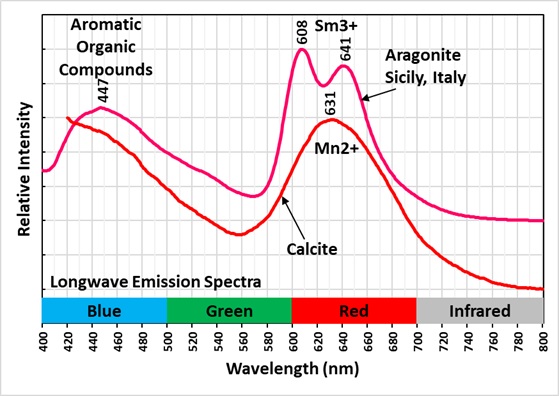
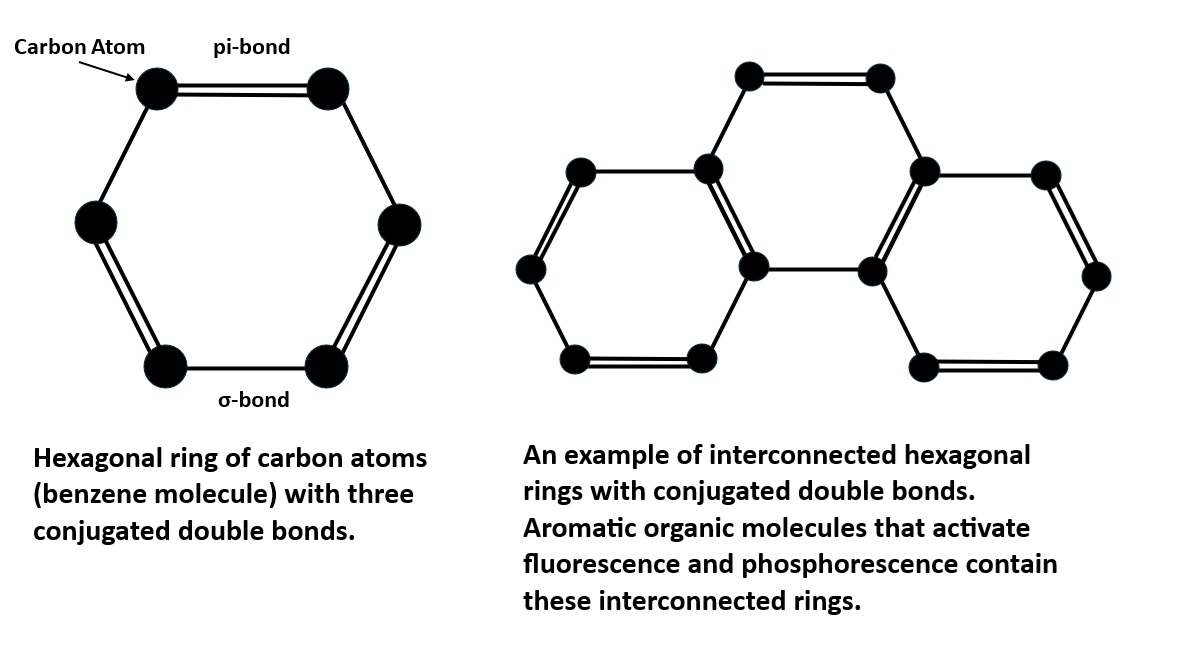
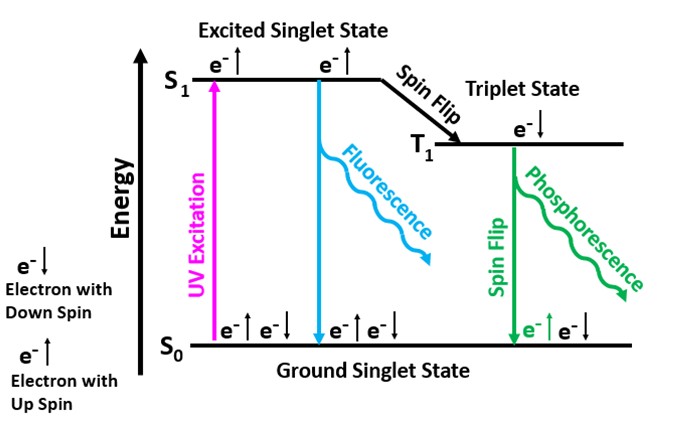
The longwave emission spectrum of this specimen indicates it has two types of fluorescent activators. The first type of activator is aromatic organic compounds. The organics cause a broad peak in the blue region with a maximum at 447 nm. It also causes long lasting green afterglow after exposure to LW light. This type of fluorescence and afterglow is referred to as a singlet-triplet process that involves electron excitation and flipping of electron spin. The aromatic molecules contain interconnected hexagonal benzene rings of carbon atoms with alternating single and double bonds. These conjugated double bands allow electrons to easily move from the ground singlet state to an excited singlet state at a higher energy level by exposure to UV light. According to quantum mechanics, an electron orbital can contain either one or two electrons in the ground singlet state. If there are two electrons in the orbital, they must have opposite spins. In aromatic molecules, the singlet state contains two electrons with opposite spins and when excited by UV light one electron moves to a higher energy level of an excited singlet state. Excited singlet states are short-lived, so most electrons return to the original ground singlet state, and the excess energy is emitted as a photon. The photon emission is a broad peak with a maximum at 447 nm.
Some electrons move from the excited singlet state to a slightly lower energy level known as the triplet state. The electron spin is flipped in the triplet state, and it now has the same spin as the electron left in the ground singlet state. Quantum mechanics forbids electrons of the same spin in the ground singlet state, so the electron in the triplet state must flip its spin to return to the ground singlet state. The electron must wait until the ambient thermal crystal lattice vibrations cause it to flip again before it can fall back down to its original state and emit a photon. The triplet state is at a lower energy level compared to the excited singlet state. Therefore, the energy of the photon emitted when the electron moves back to the ground singlet state from the triplet state is less. According to Planck's Law, lower photon energy corresponds to a longer wavelength (green) for the phosphorescence/afterglow. Temperature affects the time for the spin to flip and for the electron to return from the triplet state to its original singlet state. Cooling the crystal extends the phosphorescent time.
Organic activators also cause the bluish white fluorescence and afterglow under MW and SW light. The explanation for the change in the color of the afterglow is unknown to me.
The second type of activator is samarium (Sm3+) replacing calcium. The samarium causes two peaks in the longwave emission spectrum at 608 nm and 641 nm in the red region. The two peaks differentiate samarium activation compared to manganese activation that has a single broad peak with a maximum around 631 nm. The spectrum shown in the spectral plot is from calcite, but manganese activated aragonite spectra shown in the fluomin.org website are like this calcite spectrum.
Summary of luminescence responses:
Aragonite (Mindat) (RRUFF)
- Fluorescence under Longwave (365nm LED) UV light: Red
- Afterglow after exposure to Longwave (365nm LED) UV light: Green
- Fluorescence under Midwave (305nm LED) UV light: Pink
- Afterglow after exposure to Midwave (305nm LED) UV light: Blue
- Fluorescence under Shortwave (255nm LED) UV light: Blue
- Afterglow after exposure to Shortwave (254nm Lamp/Mercury) UV light: Blue