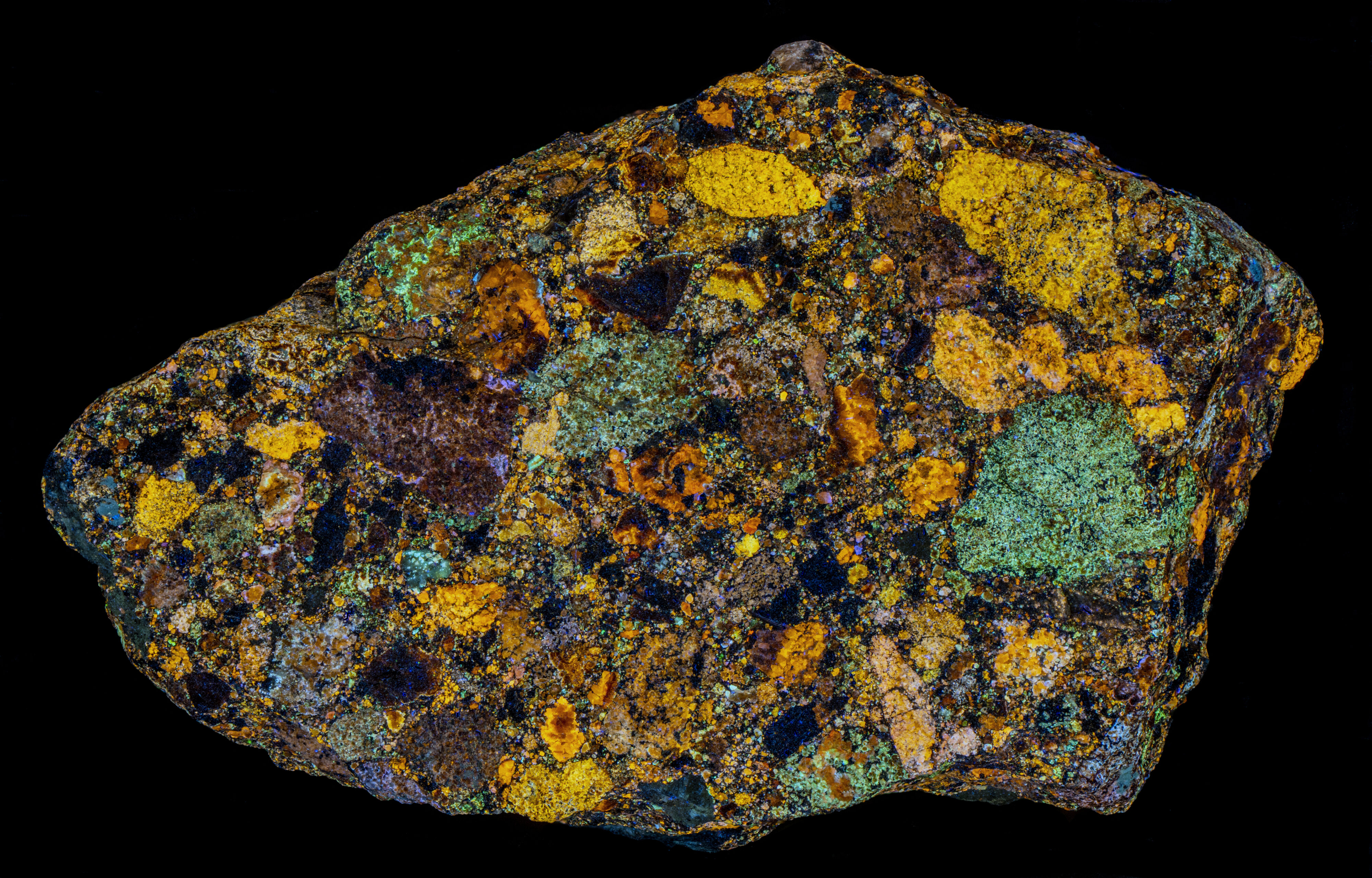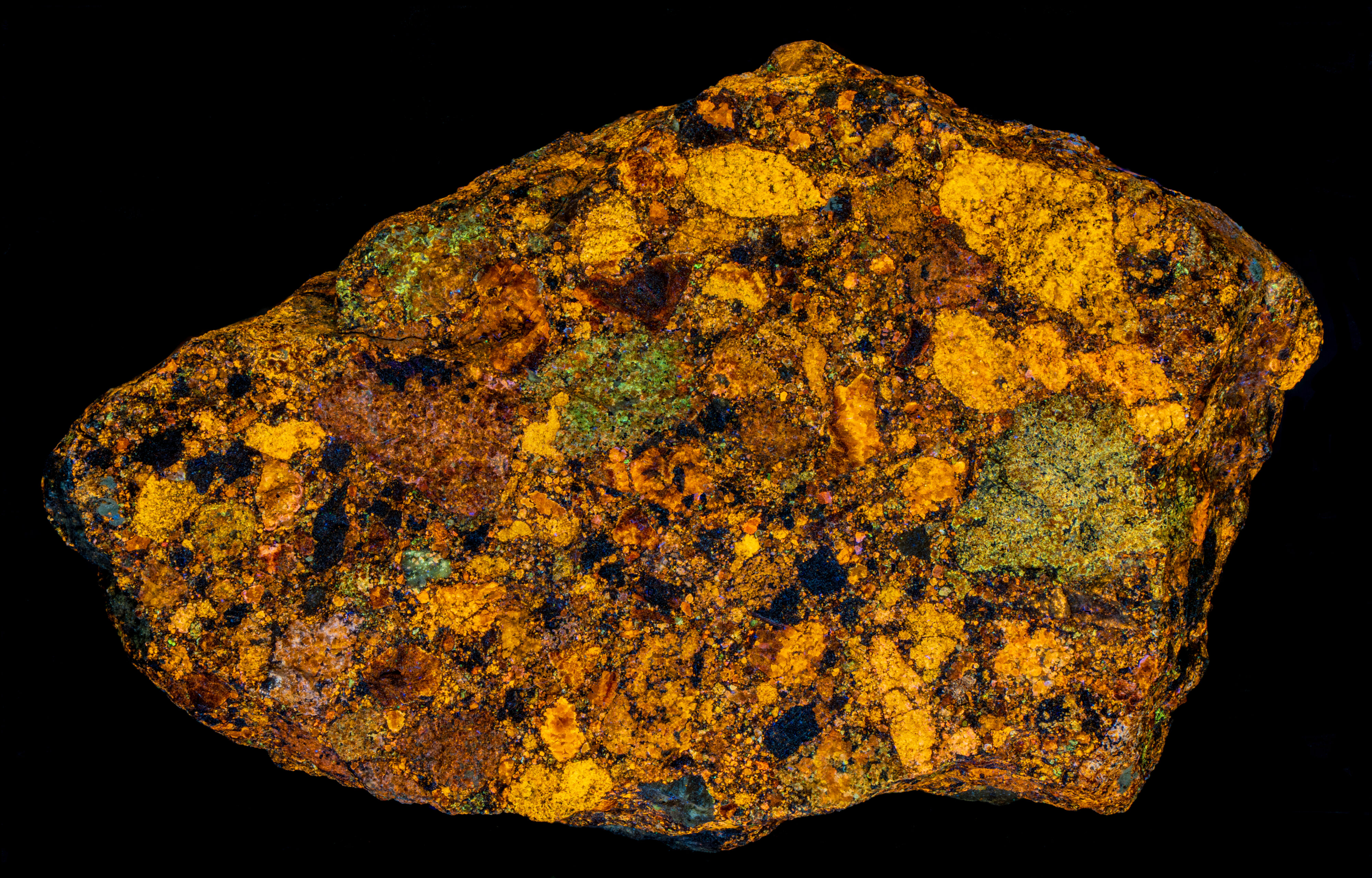Sphalerite Breccia from Horn Silver Mine, Utah
Contributed by: Michael Crawford
Date: Feb 12th, 2026
Locality: Horn Silver Mine, Frisco, San Francisco Mining District, Beaver County, Utah, USA (See on Mindat)
Size: 7 x 12.5 cm
Description:
This is a sphalerite (ZnS) specimen from the Horn Silver Mine, Beaver County, Utah. The specimen is a breccia of sphalerite fragments cemented by fine grains of sphalerite. The large fragments of sphalerite have different activators that create different fluorescent colors. Mineralization at the Horn Silver Mine is hosted by a north-south trending high angle fault zone which reaches widths of 100 feet and juxtaposes west dipping Cambro-Ordovician carbonates on its west side with Tertiary volcanics on the east side. Quartz monzonite intrudes the structure in the northern part of the mine area.
The brightest and most colorful fluorescence occurs under longwave UV illumination. The sphalerite also has long-lasting afterglow from exposure to longwave UV light. The afterglow is also multi-colored. This specimen like most Horn Silver sphalerite specimens has intense triboluminescence. It emits orange flashes when scratched with a hard object. Triboluminescence is caused by the breaking of chemical bonds and electrical charge separation. Light flashes are produced by the recombination of electrical charges.
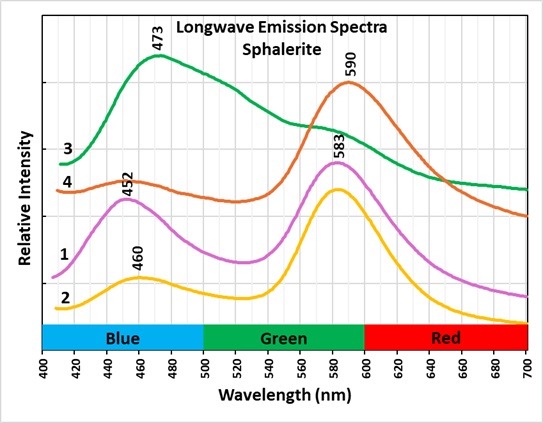
Three of the sphalerite fragments have longwave emission peaks around 583 nm to 590 nm. These peaks represent yellow to orange fluorescence and are activated by manganese replacing zinc. The peaks in the blue region around 452 nm to 460 nm may be activated by silver replacing zinc.
The longwave emission spectrum of a green sphalerite fragment (#3) is a broad asymmetric peak with a maximum at 473 nm. Green sphalerite fluorescence is caused by copper and aluminum substituting for two zinc ions. The zinc ions have a 2+ charge, copper ions have a 1+ charge, aluminum ions have a 3+ charge. Two zinc ions (2 * 2+ = 4+) are replaced by a copper ion and an aluminum ion ((1+) + (3+) = 4+) in order to maintain charge balance. Details on how the copper-aluminum replacement activates the green sphalerite fluorescence are described by Waychunas in the 2020 FMS Journal.
Summary of luminescence responses:
Sphalerite (Mindat) (RRUFF)
- Fluorescence under Longwave (365nm LED) UV light: Orange
- Fluorescence under Longwave (365nm LED) UV light: Green
- Afterglow after exposure to Longwave (365nm LED) UV light: Orange
- Afterglow after exposure to Longwave (365nm LED) UV light: Green