Fluorite from the Ojuela Mine, Mapimi, Durango, Mexico
Contributed by: Michael Crawford
Date: Sep 12th, 2025
Locality: Ojuela Mine, Mapimí, Mapimí Municipality, Durango, Mexico (See on Mindat)
Size: 5.5 x 6.5 cm
Description:
This is a specimen of purple fluorite cubes from Ojuela Mine, Mapimi, Durango, Mexico. The fluorite fluoresces bright red under longwave UV light. It’s fluorescence is dimmer under midwave light and non-fluorescent under shortwave light.
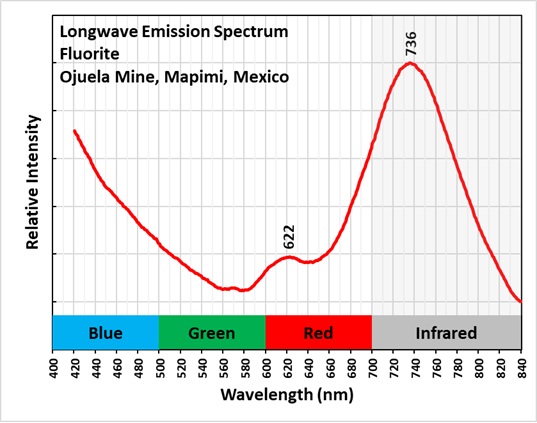
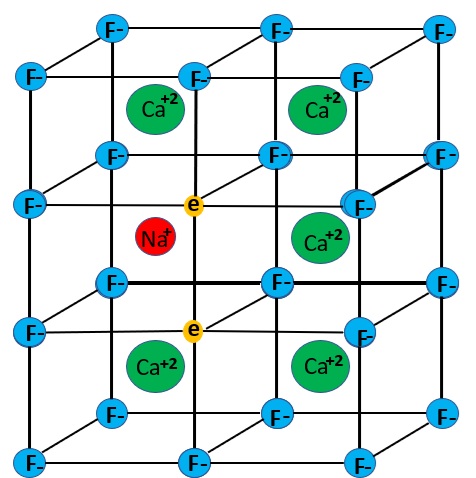
The longwave emission spectrum of the bright red fluorescence has a broad peak at 736 nm and a smaller peak at 622 nm. The emission spectra indicates that the red and near infrared fluorescence is a combination of two types of activation. Details of the two mechanisms are discussed in Gaft and others (2020). One mechanism is the presence of M-centers. An M-center forms when two nearby fluorine ions (F-) are missing and are replaced by electrons. The M-center is stabilized by replacement of a calcium ion by a sodium ion. The M-centers are formed by naturally occurring radiation that caused the formation of crystal lattice defects. The fluids that deposited the fluorite were radioactive, but there is no residual radioactivity. The radioactivity also caused the purple color of the fluorite. The purple color is caused by colloidal calcium, not the M-center. According to Gaft and others, the M-center activates a broad emission peak with a maximum at 750 nm and a smaller peak at 635 nm.
A second activator of red fluorescence is samarium (Sm2+) replacing calcium. Samarium activation produces a broad peak with a maximum at 730 nm. Emission spectra published in fluomin.org show samarium also activates a peak at 622 nm. The emission peak measured for this specimen peaks at 736 nm. The measured peak is intermediate to the 750 nm peak for the M-center and the 730 nm peak for samarium and suggests the infrared fluorescence is a mix of the two activator mechanisms. The smaller measured peak is at 622 nm. This corresponds to a samarium peak rather the 635 nm M-center peak.
Another property of red fluorescent fluorite is that prolonged exposure to longwave UV light bleaches the red fluorescence. Exposure to shortwave UV light can restore the longwave fluorescence. The number of times that the longwave fluorescence can be restored is limited.
Gaft, Michael & Waychunas, G. & Rossman, George & Nagli, Lev & Panczer, G. & Raichlin, Yosef. (2020). Red photoluminescence and purple color of naturally irradiated fluorite. Physics and Chemistry of Minerals. 47
Summary of luminescence responses:
Fluorite (Mindat) (RRUFF)
- Fluorescence under Longwave (365nm LED) UV light: Red