Scheelite and Fluorapatite from Franklin, New Jersey
Contributed by: Michael Crawford
Date: Dec 19th, 2025
Locality: Franklin Mine, Franklin, Sussex County, New Jersey, USA (See on Mindat)
Size: 6.5 x 13 cm
Description:
A specimen of molybdenum-rich scheelite (Ca(MoO4)) and fluorapatite (Ca5(PO4) 3F) with minor calcite on a dark pyroxene matrix. The specimen comes from Franklin, Sussex County, New Jersey. The scheelite fluoresces off-white under shortwave UV light and is slightly yellower under midwave UV light. The fluorapatite fluoresces orange under both shortwave and midwave light. Calcite fluoresces orange red under shortwave light. None of the minerals fluoresce under longwave light.
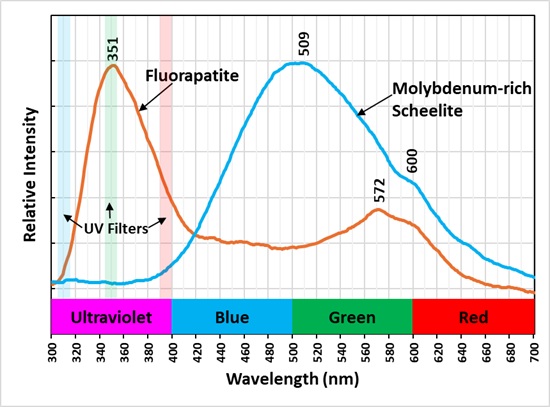
The shortwave emission spectrum of this scheelite has as broad peak with a maximum at 509 nm. Pure scheelite fluoresces light blue under shortwave light and its emission spectrum has a peak around 430 nm. The addition of molybdenum shifts the peak to longer wavelength. Scheelite (Ca(WO4)) and powellite (Ca(MoO4)) form a solid solution series where tungsten and molybdenum are readily interchangeable in the crystal structure. Shortwave scheelite fluorescence begins to change color at 1.5 molar weight percent molybdenum, and it becomes pale yellow at 10 molar weight percent molybdenum. Adding more molybdenum kept the fluorescent color the same as pure powellite. This specimen was previously featured in the Franklin-Ogdensburg Mineralogical Society (FOMS) website (https://www.fomsnj.org/mineral.aspx?minid=398&minName=Scheelite ). Scheelite from the location of this specimen contains 12 wt% molybdenum. The tungstate ion and molybdate ions are intrinsic activators for the specimen’s fluorescence. The emission spectrum also contains an inflection at 600 nm. This is likely due to a rare earth element (REE) replacing calcium and activating fluorescence with a sharp peak around 600 nm.
The shortwave emission spectrum is brightest in the ultraviolet with a maximum peak at 351 nm. This peak is likely activated by the rare earth cerium (Ce3+) replacing calcium. The emission spectrum of the orange, fluorescent color, has a double peak at 572 nm and another around 600 nm. The visible fluorescence may be activated by manganese and/or rare earth elements (REE) replacing calcium.
A false color image shows the ultraviolet fluorescence of the specimen. The narrow bandpass filters used to take pictures of the fluorescence are shown the plot of the emission spectra. The three bands were combined to make the false color image. The fluorapatite appears green and scheelite appears red in the false color image. The shoulder of the scheelite emission extends into the ultraviolet and it is detected in the 394 nm narrowband image. The 350 nm narrowband image captured the cerium activated fluorescence of the fluorapatite.
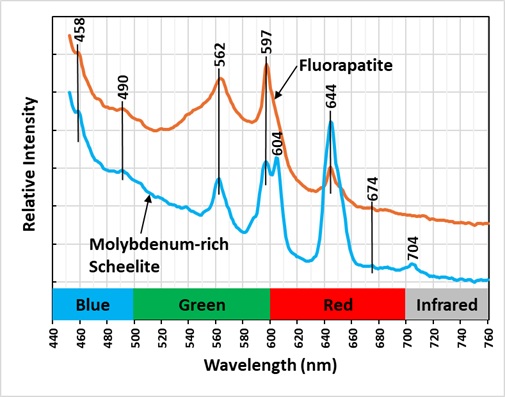
The last image shows emission spectra measured with 405 nm laser illumination. There is essentially no fluorescence with bright 365 nm LED illumination, but the laser intensity is much brighter than the LED’s and can make the scheelite and fluorapatite fluoresce with enough intensity to get a spectral measurement. The spectra contain several sharp peaks and several peaks are the same for both minerals. The major peaks in the spectra are likely activated by the rare earth element, samarium (Sm3+).
Summary of luminescence responses:
Scheelite (Mindat) (RRUFF)
- Fluorescence under Shortwave (255nm LED) UV light: White
- Fluorescence under Midwave (305nm LED) UV light: Yellow
- Fluorescence under Shortwave (255nm LED) UV light: Orange
- Fluorescence under Midwave (305nm LED) UV light: Orange