Two Color Willemite Specimen from Franklin, New Jersey
Contributed by: Michael Crawford
Date: Nov 12th, 2025
Locality: Buckwheat dump mineral collecting site, Franklin Mine, Franklin, Sussex County, New Jersey, USA (See on Mindat)
Size: 7 x 10.5 cm
Description:
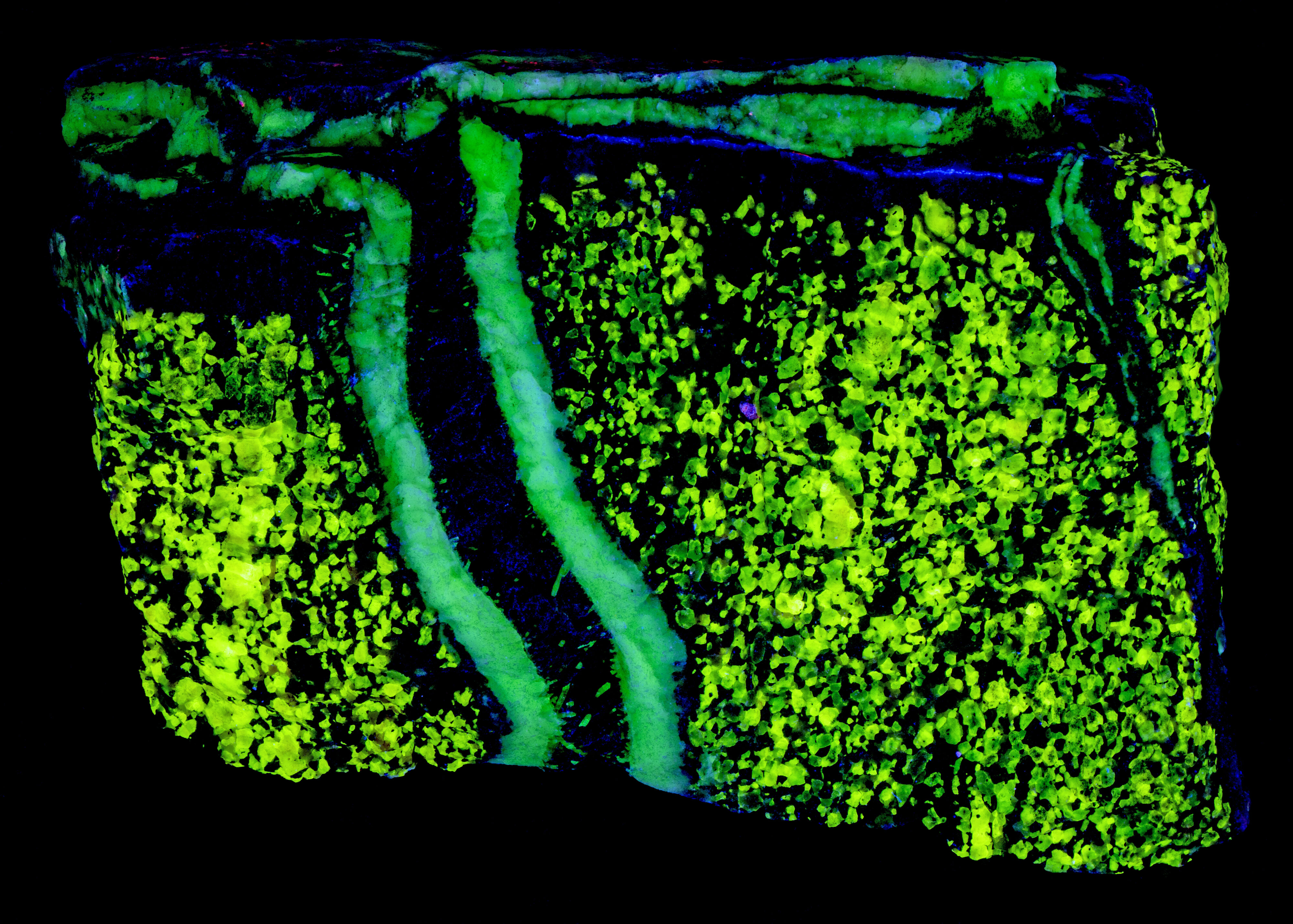
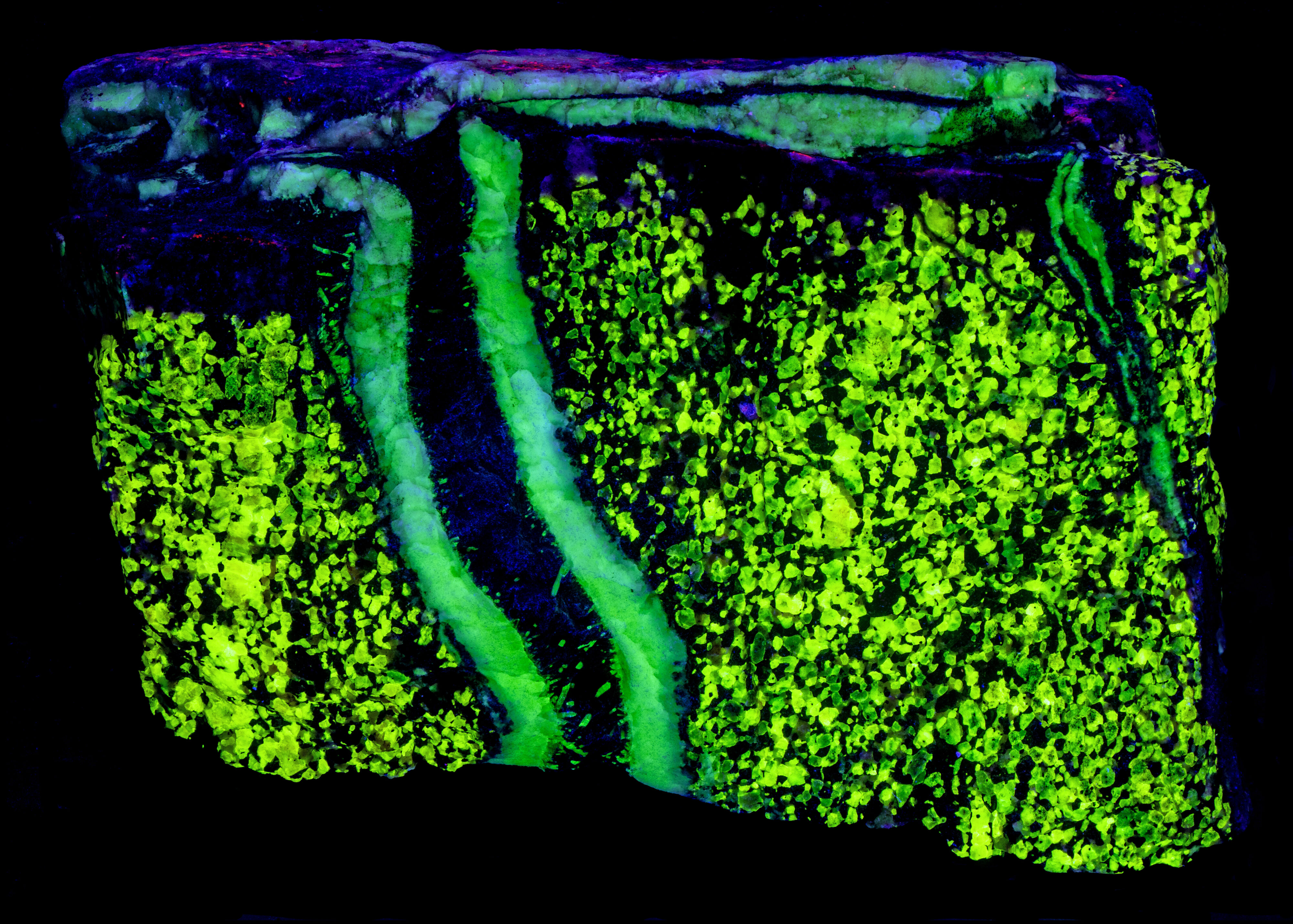
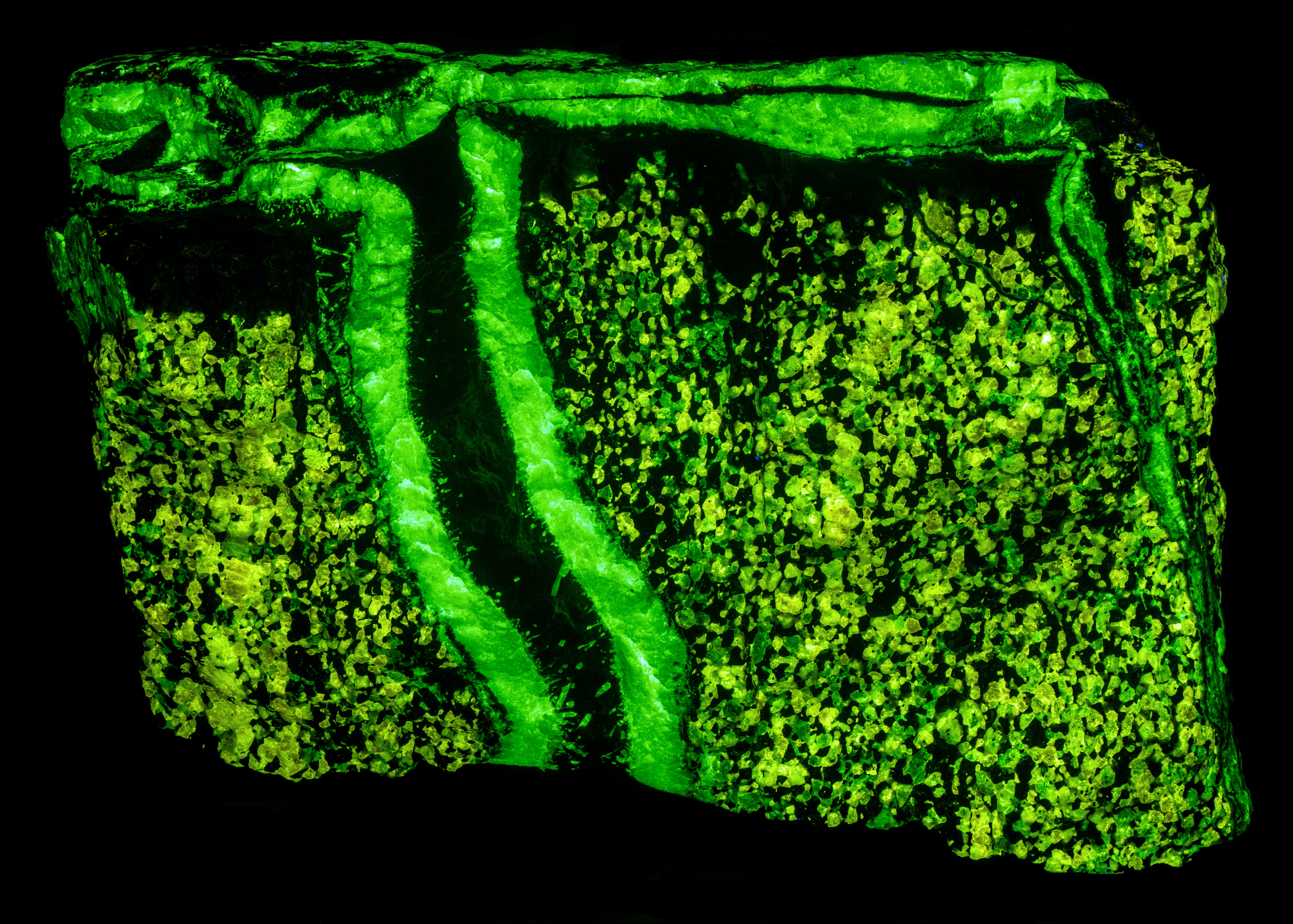
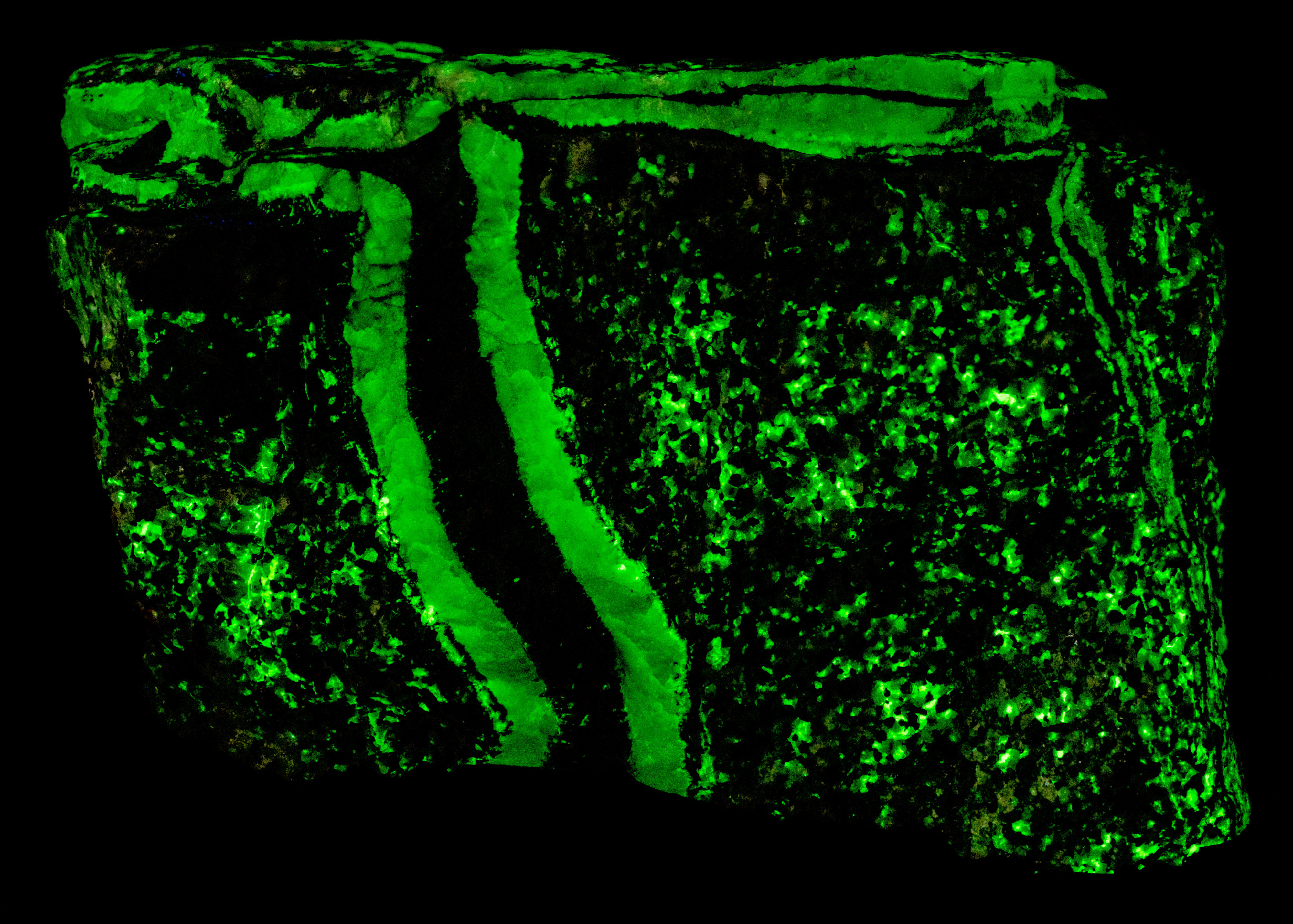
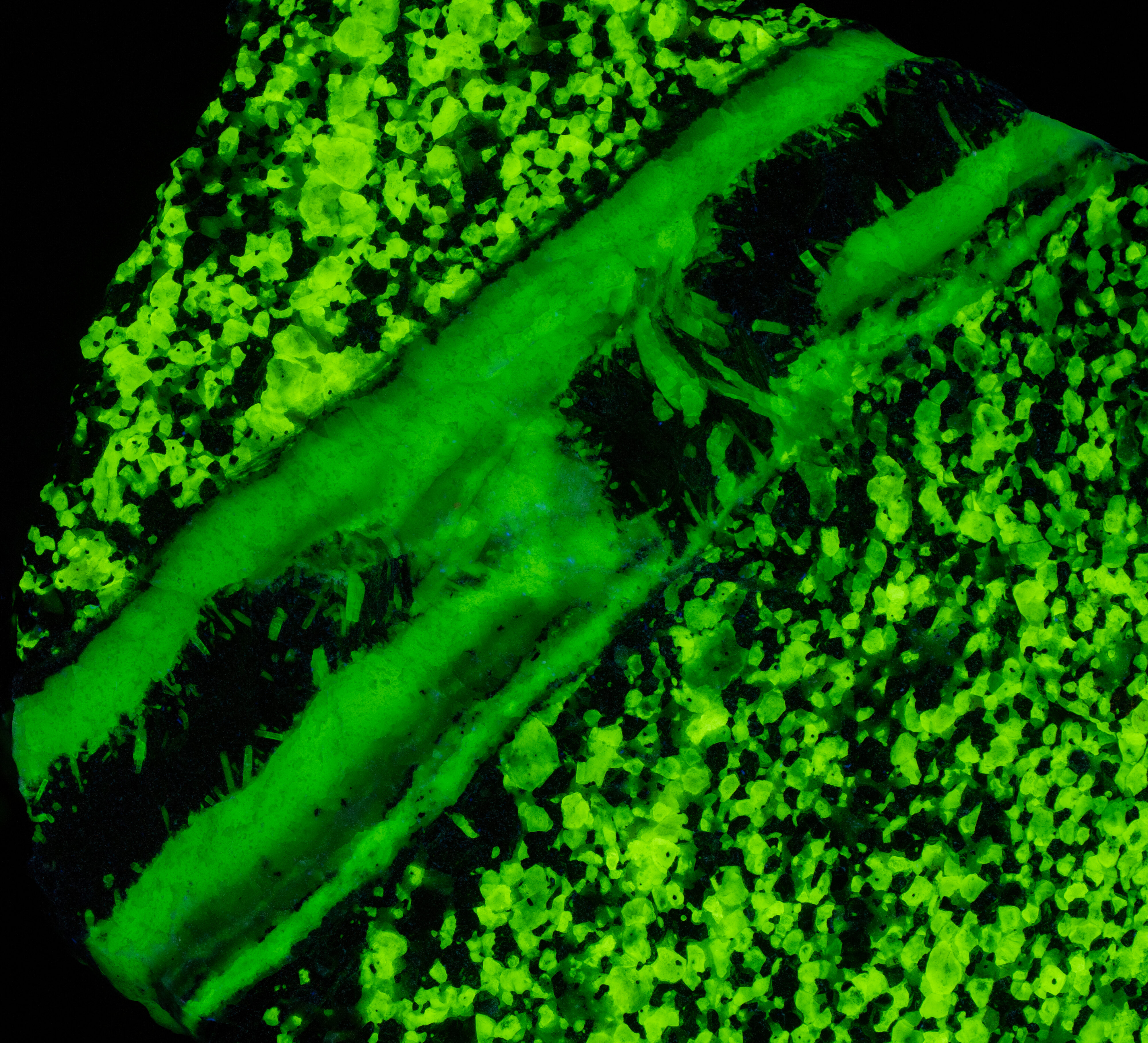
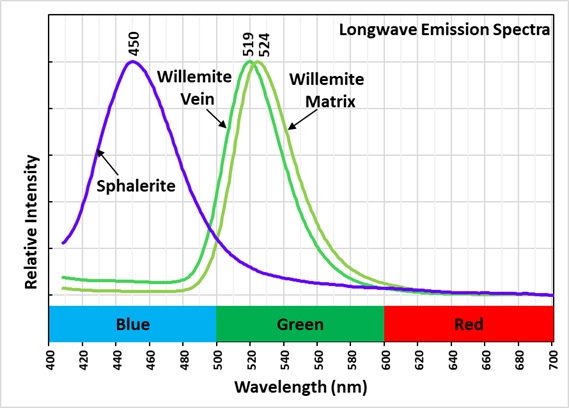
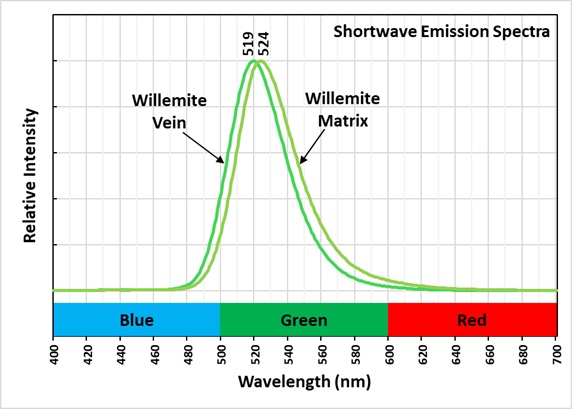
This is a two color willemite specimen from the Buckwheat Dump, Franklin, New Jersey. Willemite grains in the gneiss matrix fluoresce yellow-green compared to the greener fluorescence of the white willemite in the veins. The two types of willemite fluorescence is the same under all wavelengths of UV illumination. It is difficult to capture this fluorescent difference with a standard camera, but the difference can be seen in images taken using a magenta filter. The willemite color difference can be seen visually and in the emission spectra. Both the longwave and shortwave emission spectra show the vein willemite with a peak at 519 nm and the matrix willemite has a peak at 524 nm. The cause of this difference might be the concentration of the manganese activator replacing zinc in the willemite. Low concentration of manganese replacement of zinc creates isolated Mn2+ ions can create longer afterglow in synthetic willemite. Shorter afterglow occurs when the manganese concentration increases and there are more Mn2+ ions that are close to each other.
The willemite in the veins has long lasting afterglow from exposure to midwave and shortwave light. Some of the willemite grains in the matrix also have afterglow. There are two mechanisms of luminescence occurring while the UV light strikes the willemite, fluorescence and thermoluminescence that is responsible for the afterglow. The willemite fluoresces with electrons excited to a higher energy level and then emit green photons when the electrons quickly return to the ground state. Some of the excited electrons are “trapped” by nearby arsenic atoms that substitute for silicon. Some trapped electrons return to the ground state while the UV light is on, and some electrons can stay trapped for a long time after the UV light is turned off until there is enough thermal energy to push them out of the trap and back to the ground state of the Mn2+ atom.
Vertical veins of white willemite also contain a black unknown mineral that is non-fluorescent. It is likely a manganese bearing mineral or possibly serpentine. The black mineral does not react to acid. The franklinite in the matrix attracts a strong magnet and the black material in the vein also attracts the magnet. There are small willemite crystals that extend into the black mineral from the massive white willemite.
The vertical veins are truncated by a horizontal vein at the top of the specimen. The horizontal vein also contains a veinlet of violet, fluorescent sphalerite. Emplacement of the horizontal vein created a non-fluorescent zone in the matrix next to the vein.
Summary of luminescence responses:
Willemite (Mindat) (RRUFF)
- Fluorescence under Longwave (365nm LED) UV light: Green
- Fluorescence under Midwave (305nm LED) UV light: Green
- Fluorescence under Shortwave (255nm LED) UV light: Green