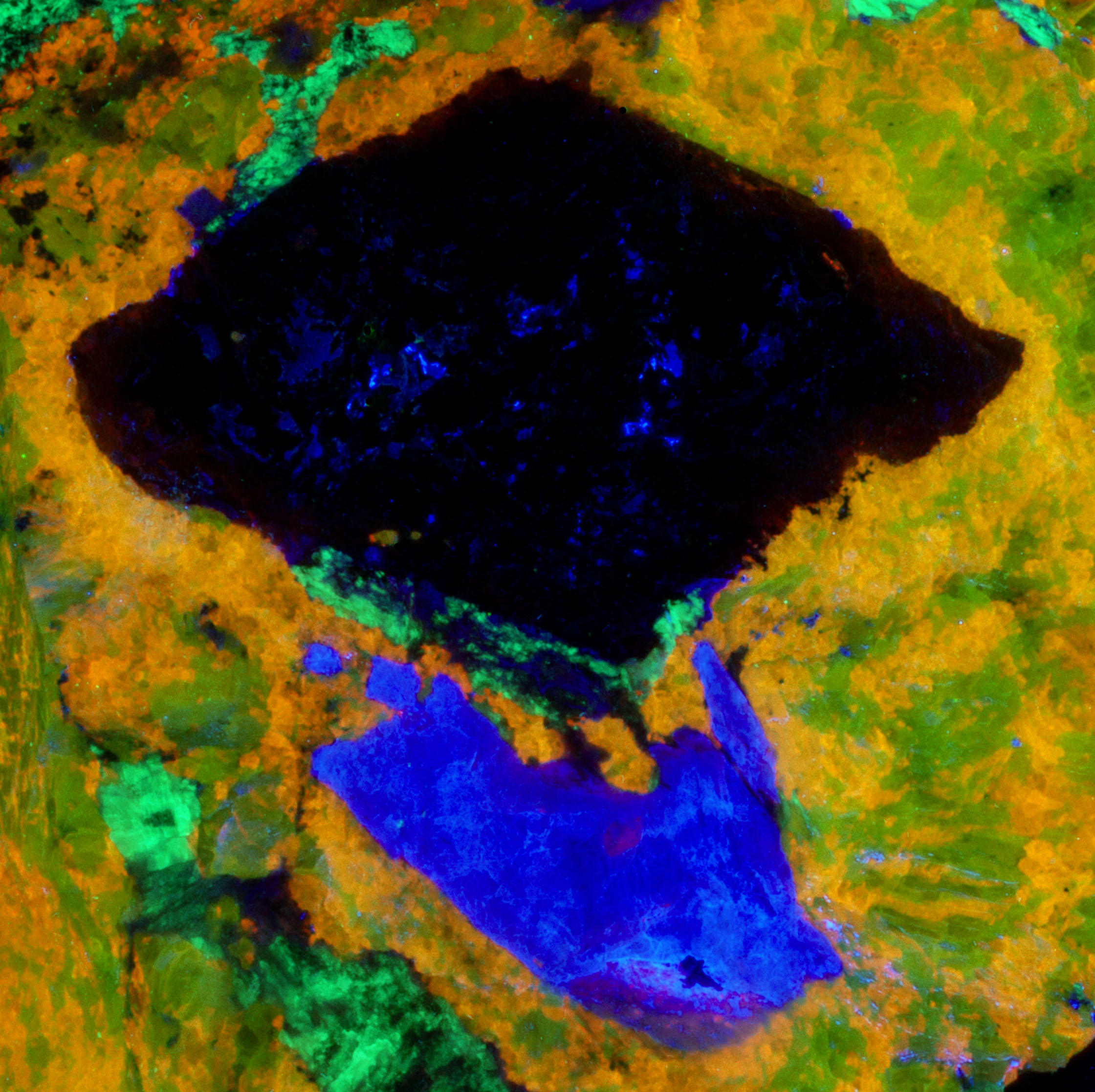
Altered Thorite, Agrellite and Two Feldspars from the Kipawa Complex, Canada
Contributed by: Michael Crawford
minID: PRN-H1C
Date: Aug 19th, 2025
Locality: Kipawa alkaline complex, Les Lacs-du-Témiscamingue, Témiscamingue RCM, Abitibi-Témiscamingue, Québec, Canada (See on Mindat)
Size: 14 x 14 cm
Description:
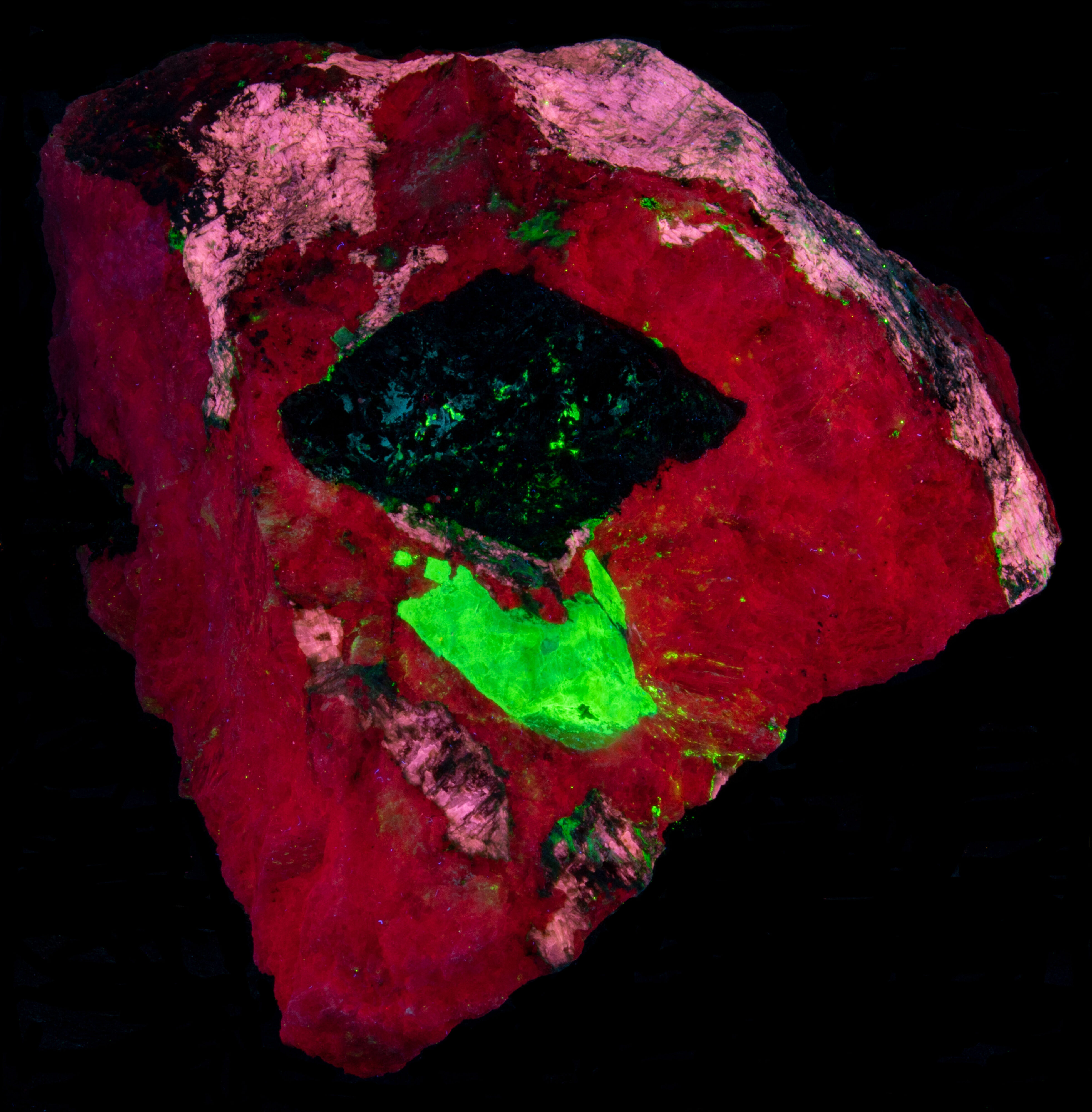
This is a specimen from the Kipawa alkaline complex, Les Lacs-du-Témiscamingue, Témiscamingue RCM, Abitibi-Témiscamingue, Québec, Canada. The specimen contains fluorescent agrellite (NaCa2Si4O10F), albite (Na(AlSi3O8)), microcline (K(AlSi3O8)) and a yellow altered crystal of thorite (Th(SiO4)). It also contains a nice red eudialyte (Na15Ca6Fe3Zr3Si(Si25O73)(O,OH,H2O)3(Cl,OH)2) crystal that is non-fluorescent and black non-fluorescent amphibole.
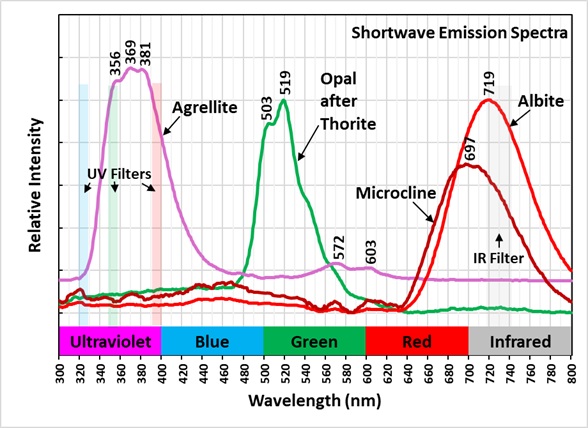
The previous owner of this specimen posted pictures of it in mindat. https://www.mindat.org/photo-525958.html and called the altered yellow crystal thorite. However, this identification is likely incorrect. Reports on XRD analyses have shown similar materials to be amorphous. The original thorite crystal has been altered by radiation destroying the thorite crystal lattice and leaving an opal pseudomorph. The original thorite likely had trace amounts of uranium in it that remained in the opal. The uranium was oxidized and formed uranyl ions that activate the green fluorescence. The altered yellow pseudomorph is radioactive. The measured total radioactivity is 1200 cpm compared to a background of 30 cpm. This radioactivity is much higher compared to most green, fluorescent opal deposited by precipitation from silica rich fluids. A commentor in a Facebook post of this specimen said that he measured a similar waxy yellow mineral in his Kipawa specimen with a gamma spectrometer and found all the radioactivity was caused by thorium. The yellow material in this specimen is unreactive to acid. Others have described green, fluorescent material from Kipawa as reactive to acid indicating calcite. The altered thorite emission spectrum exhibits two sharp peaks at 503 nm and 519 nm. This spectrum is characteristic of uranyl activation.
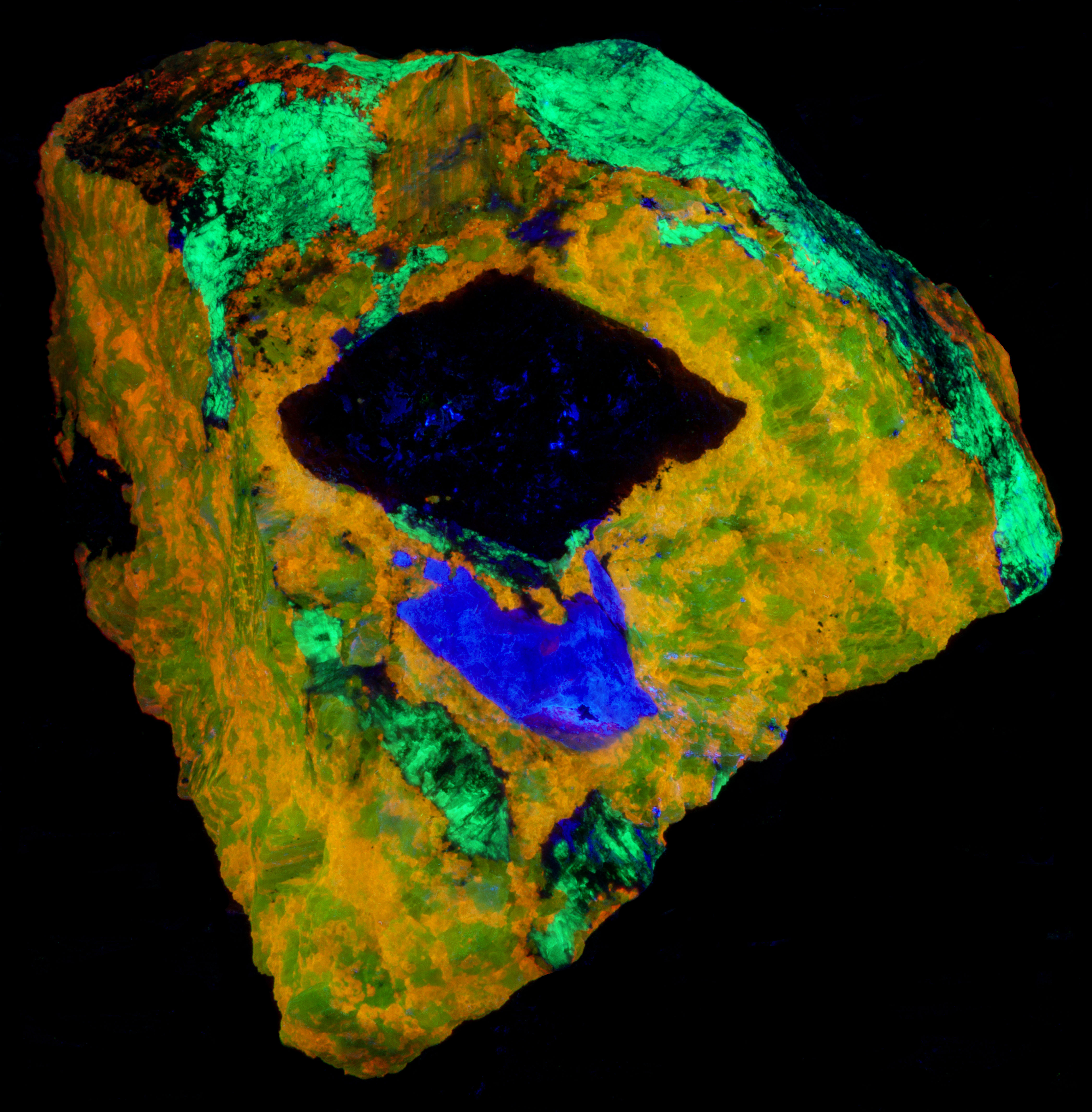
The shortwave emission peak of agrellite has several peaks that are activated by rare earths. Agrellite has very bright fluorescence in the ultraviolet that is considerably brighter than its visible fluorescence. The ultraviolet fluorescence is activated by cerium (Ce3+) that replaces calcium in the agrellite structure. Cerium causes three sharp peaks at 356 nm, 369 nm and 381 nm. There is a small peak at 572 nm that is caused by dysprosium (Dy2+) replacing calcium and there is another peak at 603 nm caused by samarium (Sm3+) replacing calcium. The false color image shows the bright ultraviolet fluorescence of the agrellite. This image is composed of three narrow band images that are shown on the spectral plot. The 310 nm band is assigned to blue, the 350 nm band is assigned to green, and the 394 nm band is assigned to red. The agrellite appears yellow in the false color image of ultraviolet fluorescence.
The near infrared pictures show two types of feldspar. In the true color image, the feldspar fluorescence is a uniform red and has been identified as albite. The near infrared image shows zoning in the feldspar. The bright areas are albite and the shortwave emission spectrum shows a peak at 719 nm. The darker areas are microcline with a shortwave emission peak at 697 nm. The albite and microcline have similar fluorescent brightness in the visible red. However, in the near infrared the microcline is dimmer, and its peak is shifted into the visible red.
Summary of luminescence responses:
Thorite (Mindat) (RRUFF)
- Fluorescence under Shortwave (255nm LED) UV light: Green
- Fluorescence under Shortwave (255nm LED) UV light: Red
- Fluorescence under Shortwave (255nm LED) UV light: Red
- Fluorescence under Shortwave (255nm LED) UV light: Pink